The EU Health Technology Assessment Regulation (EU) 2021/2282 creates a common European-level clinical assessment, the Joint Clinical Assessment (JCA), that will sit on top of, rather than replace, national pricing and reimbursement systems.
For companies developing medicines, diagnostics, and medical devices, this means that evidence generation and market access strategy must increasingly be planned from a genuinely European perspective.
What the EU HTA Regulation Does
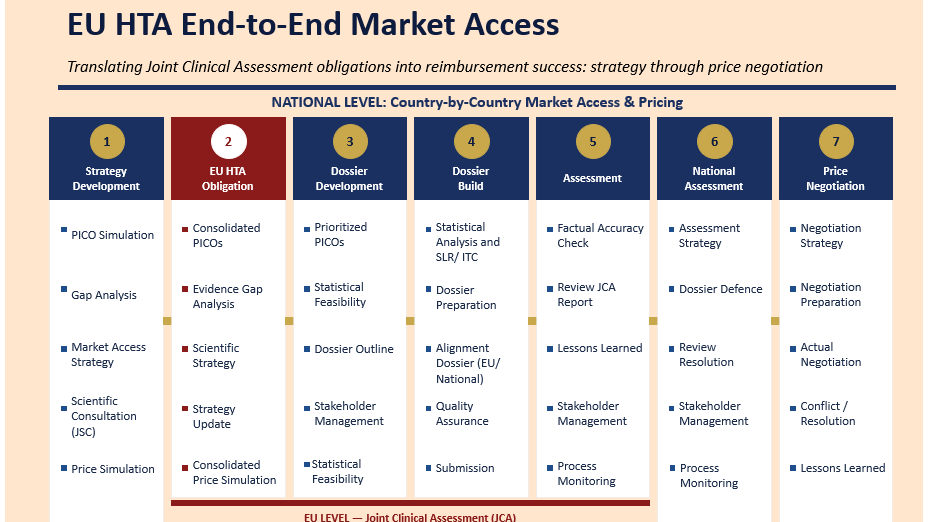
The Regulation on Health Technology Assessment (HTAR, 2021/2282) entered into force in January 2022 and has applied since 12 January 2025 across the European Union.
It establishes a permanent EU framework for:
- Joint Clinical Assessments (JCA)
- Joint Scientific Consultations (JSC)
- Identification of emerging health technologies (horizon scanning)
- Continued voluntary cooperation between national HTA bodies
The regulation’s central objective is to harmonise the assessment of relative clinical effectiveness and safety across Europe, reduce duplication of HTA work for national authorities and industry, and ultimately support faster patient access to innovative health technologies.
The Joint Clinical Assessment (JCA)
The Joint Clinical Assessment is the core mechanism of the EU HTA framework.
JCAs are EU-level scientific reports evaluating the comparative clinical effectiveness and safety of a new health technology versus relevant comparators.
These assessments are prepared collaboratively by HTA experts from multiple Member States under the coordination of the European HTA Coordination Group.
Crucially, the JCA does not determine reimbursement or pricing decisions.
Specifically, the JCA does not:
- decide reimbursement eligibility
- set prices
- conduct cost-effectiveness analysis
- perform budget-impact modelling.
Those responsibilities remain entirely within the competence of national authorities.
Instead, the JCA provides a shared clinical evidence base that Member States are expected to use as a scientific input in their national HTA and reimbursement procedures.
This avoids the need for each country to conduct a full independent clinical assessment from scratch.
The 100-Day Submission Window
One of the most significant operational features of the Joint Clinical Assessment process is the compressed submission timeline.
Once the consolidated PICO framework has been communicated to the manufacturer, companies typically have approximately 100 days to finalise and submit the JCA dossier.
This timeline is considerably shorter than many traditional national HTA submissions. Importantly, the JCA dossier must address multiple comparators, patient subgroups, and outcomes simultaneously, reflecting the evidence expectations of all participating Member States.
As a result, evidence preparation cannot begin once the final PICO is confirmed. Manufacturers must anticipate potential comparators and initiate evidence generation in advance. Without early preparation, companies risk entering the JCA process with incomplete evidence packages, which could result in explicit “no evidence available” statements in the final assessment report.
Because the JCA report is visible to all Member States, such evidence gaps could influence reimbursement negotiations across multiple European markets simultaneously.
Scope and Phasing of the EU HTA Regulation
The EU HTA system is being introduced gradually.
From January 2025, Joint Clinical Assessments are mandatory for:
- new oncology medicines
- advanced therapy medicinal products (ATMPs) authorised at EU level.
The scope will expand over time.
Planned milestones include:
- 2028 – inclusion of orphan medicines
- 2030 – all centrally authorised new medicines
- future implementation acts may extend the framework to high-risk medical devices.
This staged implementation represents a significant shift for Europe, which historically relied on fragmented country-by-country HTA processes.
The emerging architecture creates a two-tier system:
- European level – shared assessment of clinical evidence through the JCA
- National level – economic evaluation, pricing negotiations, and reimbursement decisions.
National Decision-Making Remains Central
Despite the new EU framework, national reimbursement systems remain fully sovereign.
Member States continue to determine:
- inclusion of technologies within national benefit catalogues
- cost-effectiveness analysis and willingness-to-pay thresholds
- budget-impact assessments
- pricing negotiations, discounts, and managed-entry agreements.
Because healthcare systems differ substantially across Europe, countries may still interpret and apply the JCA’s clinical findings differently.
Consequently, access conditions and reimbursement timelines will continue to vary across Member States.
Early Implementation: A System Still Taking Shape
Although the EU HTA Regulation became operational in January 2025, the system is still in its early stages of implementation.
Initial projections suggested that a larger number of technologies would enter the Joint Clinical Assessment process during the first implementation period. In practice, many manufacturers appear to have adopted a cautious approach, waiting to observe how the earliest assessments unfold before submitting their own products into the process.
As the first JCA reports are published, they will establish important methodological precedents that are likely to shape evidence strategies, comparator selection, and submission practices for future technologies entering the European market.
Strategic Implications for Market Access
The EU HTA Regulation significantly changes how companies must approach evidence generation and market access planning in Europe.
Evidence strategies can no longer be designed primarily around a small number of national HTA agencies.
Instead, clinical development programmes must satisfy a multi-country HTA audience simultaneously.
Key implications include:
- greater emphasis on appropriate comparator selection across multiple healthcare systems
- stronger need for head-to-head trials or robust indirect treatment comparisons
- increased importance of clinically meaningful endpoints and subgroup analyses.
Companies will also need closer integration between regulatory and market access teams, ensuring pivotal clinical trials satisfy both the requirements of the European Medicines Agency and anticipated evidence expectations within the JCA framework.
The Population–Intervention–Comparator–Outcome (PICO) framework defines the scope of evidence assessed within the Joint Clinical Assessment.
Under the EU HTA Regulation, PICO definitions must reflect the clinical evidence needs of multiple Member States simultaneously. This creates a significantly more complex analytical environment than traditional national HTA processes, where comparator expectations were often limited to a single jurisdiction.
Predicting likely PICO configurations has therefore become a critical strategic capability for manufacturers. Companies increasingly rely on structured HTA landscape analyses, systematic literature reviews, and expert consultation across multiple European healthcare systems to anticipate potential comparator requirements.
Because industry is not formally involved in the final PICO scoping decision, early modelling of potential scenarios can help identify evidence gaps and guide the development of indirect treatment comparisons or real-world evidence strategies before the formal JCA timeline begins.
Joint Scientific Consultations (JSC)
To support this new system, the EU HTA Regulation also introduced Joint Scientific Consultations.
JSCs provide developers with early coordinated advice from European HTA bodies on clinical development programmes.
These consultations can address:
- choice of comparators
- endpoint selection
- study design
- target patient populations.
Joint Scientific Consultations can also run in parallel with EMA scientific advice, helping align regulatory and HTA evidence expectations.
Initial JSC rounds began prior to the 2025 implementation and continue through 2026, with several consultations already completed.
This indicates that the new system is now fully operational and increasingly used by industry.
Key Takeaways for Manufacturers
The EU HTA Regulation introduces several important strategic implications for companies developing health technologies.
Think European
Clinical trials and evidence packages should increasingly be designed with EU-level Joint Clinical Assessment requirements in mind, rather than focusing exclusively on individual national HTA agencies.
Invest in Early Scientific Advice
Joint Scientific Consultations and parallel EMA–HTA advice can significantly reduce evidence gaps and improve alignment between regulatory approval and reimbursement evaluation.
Build National Value Narratives from a Shared Clinical Base
While the JCA will provide a common clinical evidence foundation, companies will still need to develop country-specific economic and value narratives that reflect national healthcare priorities and budget constraints.
Expect Faster but More Rigorous Scrutiny
Although the JCA should reduce duplication of clinical assessments across Europe, it will also introduce greater methodological scrutiny at the EU level.
If a technology receives an uncertain or negative clinical assessment within the JCA, that conclusion may influence multiple national reimbursement processes simultaneously.
Frequently Asked Questions
1. How does the Joint Clinical Assessment differ from traditional national HTA evaluations?
The Joint Clinical Assessment (JCA) evaluates the relative clinical effectiveness and safety of a new health technology compared with appropriate standard-of-care comparators across Europe.
Traditionally, national HTA bodies such as the Haute Autorité de Santé or the Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen would independently analyse clinical trial evidence, often applying different comparator assumptions and methodological frameworks.
Under the EU HTA Regulation, the JCA produces a single shared scientific report on clinical outcomes, typically analysing endpoints such as overall survival, progression-free survival, quality-of-life measures, and safety signals across relevant subgroups.
National HTA bodies will then use this shared clinical evidence as a basis for their own economic and reimbursement assessments, rather than repeating full clinical evaluations.
2. Does the EU HTA Regulation harmonise reimbursement decisions across Europe?
No. The EU HTA Regulation explicitly does not harmonise pricing or reimbursement decisions.
Member States retain full responsibility for:
- health-economic evaluation (cost-effectiveness modelling, budget impact analysis)
- willingness-to-pay thresholds
- inclusion of technologies in national benefit catalogues
- pricing negotiations and managed entry agreements.
This reflects the principle that healthcare financing remains a national competence within the European Union.
Consequently, even with a shared Joint Clinical Assessment, countries may still reach different conclusions regarding reimbursement eligibility based on national healthcare priorities and budget constraints.
3. What types of clinical evidence will be most influential in Joint Clinical Assessments?
The EU HTA framework places strong emphasis on comparative evidence demonstrating relative clinical benefit.
Evidence likely to be particularly influential includes:
- head-to-head randomised controlled trials versus standard of care
- robust indirect treatment comparisons or network meta-analyses
- clinically meaningful endpoints such as overall survival or patient-reported outcomes
- subgroup analyses relevant to heterogeneous patient populations.
Because the JCA must support decision-making across multiple healthcare systems, comparators must reflect widely accepted standards of care across Europe, which may differ from those used in individual national HTA processes.
4. How do Joint Scientific Consultations help developers align regulatory and HTA evidence requirements?
Joint Scientific Consultations (JSCs) allow developers to obtain early methodological advice from European HTA bodies regarding their clinical development programmes.
These consultations typically address questions such as:
- appropriate comparator therapies
- endpoint selection
- study design and population selection
- duration of follow-up required to demonstrate clinical benefit.
JSCs may be conducted in parallel with scientific advice from the European Medicines Agency, helping companies align regulatory approval requirements with reimbursement evidence expectations.
This early dialogue can significantly reduce the risk that pivotal clinical trials will later be considered insufficient for HTA evaluation.
5. How might the EU HTA Regulation affect market access timelines for innovative technologies?
In theory, the EU HTA Regulation should reduce duplication of clinical assessments across Member States, potentially accelerating access to innovative medicines.
However, the system may also introduce greater methodological scrutiny at the European level.
If the Joint Clinical Assessment concludes that a technology has uncertain or limited comparative benefit, that conclusion could influence multiple national HTA decisions simultaneously.
As a result, while clinical evaluation may become more efficient, the stakes associated with the JCA process are considerably higher, making early evidence planning and comparator strategy critical for successful European market access.
References
- European Union.
Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on Health Technology Assessment.
Official Journal of the European Union. 2021.
https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32021R2282
- European Commission.
Health Technology Assessment Regulation: Implementation and Operational Framework.
European Commission Directorate-General for Health and Food Safety (DG SANTE).
https://health.ec.europa.eu/health-technology-assessment_en
- European Medicines Agency.
EU HTA Regulation and interaction with the EMA regulatory framework.
EMA policy brief and guidance materials.
https://www.ema.europa.eu
- Schuster V, et al.
EU HTA Regulation and Joint Clinical Assessment: Threat or Opportunity for Market Access?
Journal of Market Access & Health Policy. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11130920/
- Johns A.
EU Joint Clinical Assessment: A Framework for Optimising Health Technology Assessment Cooperation.
Journal of Market Access & Health Policy. 2025.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12551043/
- Desmet T, et al.
Implementing the EU HTA Regulation: Stakeholder Perspectives and Implications for Market Access.
Frontiers in Pharmacology. 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11039851/
- EUnetHTA.
Methodological Guidelines and Final Joint Action Reports.
European Network for Health Technology Assessment.
https://www.eunethta.eu
- Monnereau M, Jarne A, Benoist A, Fradet C, Perol M, Filleron T, Baschet L.
Methodological Advances and Challenges in Indirect Treatment Comparisons for Health Technology Assessment.
Health Economics Review. 2025.
- Claxton K, Sculpher M, Culyer A, et al.
Methods for the Estimation of the National Institute for Health and Care Excellence Cost-Effectiveness Threshold.
Health Technology Assessment. 2015.
- Towse A, Barnsley P.
Approaches to Value Assessment for Innovative Medicines and Technologies in Europe.
Office of Health Economics. 2021.
- Chalkidou K, Levine R, Dillon A.
Helping Poor Countries Make Health Decisions: NICE International and the Globalisation of HTA.
Health Affairs. 2010.
- Wild C, Stricka M, Patera N.
Health Technology Assessment in Europe: Current Landscape and Future Development.
Austrian Institute for Health Technology Assessment (AIHTA). 2023.
- World Health Organization.
Health Technology Assessment: Global Policy Frameworks and Methods.
WHO Technical Report Series.
https://www.who.int
