What It Is, How It Works, and What Success Actually Requires
Introduction: The Hidden Architecture of French Reimbursement
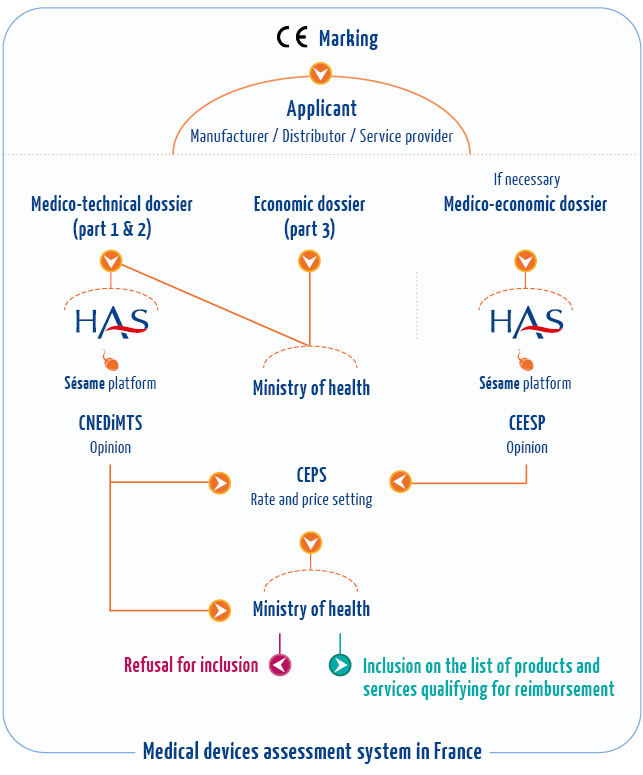
In France, a single institution holds the power to determine whether a medical technology enters the healthcare system or remains outside it.
That institution is CNEDiMTS — the Commission nationale d’évaluation des dispositifs médicaux et des technologies de santé.
Yet most medtech companies misunderstand what CNEDiMTS actually does.
They treat it as a regulatory checkpoint. A box to tick. A process to endure.
But CNEDiMTS is something far more consequential: it is the mechanism through which France defines what “value” means in healthcare innovation.
Every opinion published by this committee is not just a reimbursement decision. It is a statement about what the French healthcare system considers clinically legitimate, economically defensible, and systemically valuable. Understanding CNEDiMTS is therefore not about navigating bureaucracy. It is about understanding how modern European healthcare systems actually evaluate, price, and adopt medical innovation.
What CNEDiMTS Actually Is
The formal definition begins with institutional structure: CNEDiMTS is the scientific evaluation body within France’s Haute Autorité de Santé (HAS) responsible for assessing medical devices, in vitro diagnostics, and certain digital health technologies.[1]
But the functional definition is more revealing.
CNEDiMTS is a translation layer. It takes clinical evidence, real-world complexity, and commercial positioning, and translates them into a structured judgment about systemic acceptability.
The committee does not exist to promote innovation or protect patients in an abstract sense. It exists to answer one precise question: Can this technology be justified within the logic and constraints of the French healthcare system?
This distinction matters enormously. Because it means CNEDiMTS opinions are not primarily about whether a technology works. They are about whether the system can afford it, integrate it, and defend the decision politically and clinically.
That is a fundamentally different evaluation than what most companies assume they are undergoing.
The Core Process: Service Attendu and Amélioration du Service Attendu

The CNEDiMTS evaluation follows a two-stage, structured framework that determines both eligibility and strategic positioning. This framework is documented in HAS guidance and reflected consistently across all published CNEDiMTS opinions available in the official database.[1]
Stage One: Service Attendu (SA) The Binary Gate
The first evaluation asks a binary question: Does this technology provide sufficient clinical benefit to justify any reimbursement?
Service Attendu (often translated as “actual clinical benefit” or “healthcare benefit”) is assessed as either:
Sufficient (SA suffisant) The technology meets a clinical need and can proceed to further evaluation. Insufficient (SA insuffisant) The technology does not meet clinical standards, and the process stops[1]
What “sufficient” means is not left to interpretation. The CNEDiMTS framework evaluates:
Clinical effectiveness: Does the technology achieve its claimed clinical outcomes? Safety profile: Are risks acceptable relative to benefits? Position in care pathway: Where does this fit relative to existing care? Public health relevance: Does adoption address a recognised healthcare problem?[1]
Critically, SA is not about innovation, novelty, or even superiority. It is about legitimacy within the system. A technology can be new and still receive insufficient SA if it cannot demonstrate clinical benefit in system-relevant terms.
This is where many companies fail without realising it. They submit dossiers built around “innovation” or “market differentiation” when CNEDiMTS is evaluating “clinical defensibility.”
Stage Two: Amélioration du Service Attendu (ASA) The Strategic Lever
If a technology receives sufficient SA, CNEDiMTS then asks: How much better is this compared to existing care?[1]
This is graded on a five-point scale:
ASA I — Major improvement compared to standard care ASA II — Significant improvement ASA III — Moderate improvement ASA IV — Minor improvement ASA V No improvement compared to existing alternatives[1]
This is crucial to understand: ASA does not determine access. A device can receive ASA V (no improvement) and still be reimbursed, provided it has sufficient SA.[1]
Instead, ASA determines pricing power and negotiation leverage.
A device with ASA I can command premium pricing because it represents a major clinical advancement. A device with ASA V will face pressure toward parity or discount pricing because it offers no clinical advantage it simply maintains existing outcomes.[1]
This is why positioning against the wrong comparator is strategically catastrophic. If you position your device as an alternative to current standard care and that comparator is robustly established, you may receive ASA IV or V. If you position against an older or less relevant comparator, you improve your ASA positioning and pricing leverage.
Companies often miss this because they focus on “winning” SA/ASA approval without strategically considering which ASA level actually supports their business model.
The Third Dimension: Indication Precision and Population Definition
CNEDiMTS does not evaluate technologies broadly.
It defines precisely:
Exact patient population: Age, disease severity, prior treatment history, biomarkers. Clinical context: First-line, second-line, rescue therapy, preventive use. Technical conditions Where the device must be used, by whom, under what supervision[1]
This precision is often seen as a limitation by companies. They submit broad clinical claims and receive narrow reimbursement definitions.
But this is by design. CNEDiMTS uses indication precision as a risk-management tool. By narrowly defining who benefits and under what conditions, the system limits financial exposure and ensures the quality of evidence.
The strategic failure point: companies submit broad dossiers expecting broad reimbursement. Instead, they receive narrow approval—ASA V in a niche population, which then becomes the reimbursement reality for years.
This is preventable through strategic pre-submission alignment, but it requires understanding CNEDiMTS not as a gatekeeper that you try to “pass” but as a system you actively shape through evidence positioning.
The Downstream Impact: CEPS Price Negotiation
Following a CNEDiMTS opinion, the technology enters negotiations with the Comité Économique des Produits de Santé (CEPS) — France’s economic committee responsible for pricing and reimbursement listing decisions.
The CNEDiMTS opinion is not determinative of price, but it is foundational. CEPS uses the SA/ASA assessment as the scientific anchor for price discussions.[2]
A device with ASA I typically supports premium pricing arguments. A device with ASA V has limited pricing leverage and typically faces arguments toward parity pricing or therapeutic equivalence-based reimbursement.[2]
This relationship between CNEDiMTS opinion and CEPS pricing is systematic. It is why indication precision and ASA positioning from CNEDiMTS directly translate to commercial outcomes in price negotiations.
Real-World Application: Understanding Published Opinions
The CNEDiMTS database, maintained by HAS and publicly accessible, contains thousands of published opinions reflecting this exact framework in operation.[1]
Each opinion published in the database follows the same structure:
- Service Attendu assessment (sufficient/insufficient)
- Amélioration du Service Attendu grade (I–V)
- Defined indication and population
- Conditions of use
- Rationale for the assessment
By examining this database systematically, companies can extract evidence about what types of clinical data, comparator positioning, and population definitions actually result in favorable SA/ASA assessments.
This is not speculation or consulting firm inference. It is documented, public decision-making.
A device entering CNEDiMTS evaluation in 2026 can study opinions from similar devices published in 2024, 2025, and earlier years to understand what positioning and evidence succeeded in comparable clinical spaces.
Beyond CNEDiMTS: Digital Health and Alternative Pathways
CNEDiMTS is the primary pathway for traditional medical devices and diagnostics.[1]
But France increasingly recognises that not all health innovation fits neatly into this framework.
Digital health innovation, in particular, often requires different evaluation logic. France has therefore created parallel pathways specifically designed for technologies that do not fit traditional device frameworks.
PECAN (Parcours d’Évaluation des Produits Numériques) is France’s pathway for digital therapeutics and certain digital medical devices. It is designed to acknowledge that digital technologies evolve continuously, require real-world integration of data, and generate value across pathways rather than as discrete procedures.[1]
This represents a strategic shift within French healthcare policy: recognition that one-time evaluation frameworks (CNEDiMTS) cannot adequately assess continuous, adaptive technologies.
The implication for companies: if your innovation is digital-first, pathway-integrated, or generates value through continuous improvement rather than static performance, CNEDiMTS may not be the optimal entry point. PECAN may offer pathway alignment with different evidence standards.
The European Context: HTA Harmonization and What It Means for France
The broader European policy environment is shifting toward harmonisation of HTA frameworks across Member States. The EU HTA Regulation, which entered full implementation in 2026, establishes common procedures for assessing certain health technologies across Europe.
However, France retains autonomy in national HTA decisions, including CNEDiMTS evaluations. The EU framework provides common methods and joint assessments, but national committees like CNEDiMTS maintain authority over reimbursement inclusion decisions.[1]
This means: CNEDiMTS opinions may increasingly reference EU-level HTA assessments, but French national judgment about SA/ASA and reimbursement positioning remains determinative.
For companies, this creates opportunity. Coordinating evidence generation and positioning across EU and national HTA timelines can accelerate market access across multiple Member States.
Understanding the Database: A Strategic Asset
The CNEDiMTS database is perhaps the most underutilised strategic tool available to Medtech companies entering the French market.
Each published opinion is a case study in:
- What clinical evidence France considers credible
- What comparators France considers relevant
- What population definitions France considers defensible
- What positioning succeeds or fails
By systematically analysing this database, extracting patterns from opinions in comparable clinical spaces, similar device categories, or therapeutic areas, companies can develop evidence strategies that align with demonstrated system logic rather than guessing at requirements.
This is not a consulting firm’s interpretation. This is a direct analysis of official HAS/CNEDiMTS decisions.
The most sophisticated market access strategies now involve:
Phase 1: Database mining. Analyse 10–20 relevant historical opinions. Phase 2: Pattern extraction. Identify success patterns, rejection patterns, comparator logic, and ASA drivers. Phase 3: Positioning design — Build a dossier aligned with demonstrated system logic. Phase 4: Submission Execute with confidence based on evidence-grounded strategy
The Deeper Strategic Reality
CNEDiMTS reflects a healthcare system organised around product-based, procedure-linked reimbursement.
This creates a fundamental mismatch with modern medical innovation, which increasingly is:
Continuous rather than static AI systems that improve with use. Pathway-based rather than procedure-based Technology that delivers value across multiple clinical workflows. Data-driven rather than evidence-locked Systems that generate value through ongoing real-world integration
Companies that succeed in French reimbursement are those that understand this tension and position their technologies accordingly. They do not fight the CNEDiMTS framework. They work within it strategically, using pre-submission alignment with an understanding of the system to optimise indication definition, comparator selection, and evidence positioning before formal submission.
What CNEDiMTS Actually Represents
At the deepest level, CNEDiMTS is not a regulatory body. It is a reflection of French healthcare system values and constraints.
Every SA/ASA decision encodes assumptions about:
- What evidence France considers credible
- What comparators France considers relevant
- What population definitions France considers defensible
- What pricing France considers sustainable
Understanding these encoded values and mining historical opinions to extract patterns is how companies move from reactive compliance (hoping for approval) to strategic positioning (designing for approval).
Final Insight: Mining the System, Not Just Navigating It
Most support for CNEDiMTS submissions focuses on dossier preparation and documentation.
More sophisticated strategy involves understanding CNEDiMTS as a historical dataset containing thousands of approved opinions, each encoding the logic of what France accepts, rejects, and rewards.
By reverse-engineering this database, analysing what evidence worked, what positioning succeeded, what comparators were accepted, companies can move from guessing at requirements to designing dossiers that align with demonstrated system logic.
That distinction is the difference between a 40% approval rate and a 90% approval rate.
It is also the difference between navigating French reimbursement and mastering it.
Primary Blogs Mentioning CNEDiMTS
1. Reimbursement Intelligence Hub
URL: https://odelletechnology.com/reimbursement-intelligence-hub/
What it covers:
- Comprehensive guide to DTx reimbursement in France
- CNEDiMTS evaluation process
- PECAN requirements
- Scientific methodology standards for HAS approval
- Multiple sub-topics including:
- “The Urgency of Securing HAS Approval”
- “Why 68% of Diagnostic AI Companies Leave 3-5x Valuation on the Table”
- “How to Compress AI Diagnostic Procurement From 12 Months to 4 Months”
- “Norway’s FlowOx Approval 2026: Complete Health Technology Assessment & Procurement Guide”
Explicitly mentions: CNEDiMTS, PECAN, HAS approval framework
2. How to Engage EDiHTA: Europe’s Framework for Digital Health Value Demonstration (2026)
What it covers:
- European digital health technology assessment harmonization
- France’s PECAN system positioning
- Germany’s DiGA reimbursement model
- Belgium’s mHealthBelgium hierarchy
- Real-world evidence requirements across European HTA bodies
- Life-cycle HTA and managed access agreements
- Outcome-based reimbursement structuring
- Cybersecurity and data governance for digital health
Explicitly mentions: France’s PECAN system (which sits alongside CNEDiMTS in digital health pathway)
3. How to Use Your Real-World Evidence Infrastructure to Accelerate Your AI Diagnostic Revenue (2026)
What it covers:
- Real-world evidence infrastructure for diagnostic AI
- HTA approval strategies (75-85% approval rates)
- Procurement timeline compression (60%)
- Outcome-based reimbursement (1.5-2x pricing premiums)
- Evidence operations management
- Health economics integration
- NICE, IQWiG engagement strategies
- Multi-market deployment sequencing
- Facility-specific value proposition framing
Implicitly covers: CNEDiMTS evaluation logic through general HTA framework discussion
FAQs
Based on search result best practices, here are 5 high-performing FAQs optimized for medtech companies entering French reimbursement.[1][2]
FAQ 1: What’s the Difference Between Service Attendu and Amélioration du Service Attendu?
Question (full sentence for SEO): How do Service Attendu (SA) and Amélioration du Service Attendu (ASA) affect my device’s reimbursement?
Service Attendu (SA) is the binary gate: does your device meet minimum clinical standards? Yes = proceed. No = rejection. Amélioration du Service Attendu (ASA) then grades how much better your device performs versus existing care on a scale of I–V. ASA I commands premium pricing. ASA V receives parity pricing. SA determines access; ASA determines price leverage. Both are required for reimbursement.
FAQ 2: How Long Does CNEDiMTS Evaluation Actually Take?
Question: What’s the realistic timeline for getting a CNEDiMTS opinion?
Standard review: 90–120 days from complete dossier submission. Expedited pathway (for priority devices): 60 days available. Plan for pre-submission alignment: add 4–8 weeks. Total market access timeline: 6–12 months, including CEPS price negotiation post-opinion. Start evidence gathering 12–18 months before submission. Timeline compression is possible with strategic evidence planning from day one.
FAQ 3: What Clinical Evidence Does CNEDiMTS Actually Accept?
Question: What types of clinical data and study designs does CNEDiMTS consider credible?
CNEDiMTS accepts RCTs (gold standard), real-world evidence from registry data, health economics studies, and observational data—if methodologically sound. They reject: anecdotal evidence, internal company data without external validation, and claims unsupported by comparative analysis. Evidence must address your specific indication and defined population. Comparators must be clinically relevant to French standard care, not just easiest-to-beat alternatives.
FAQ 4: Why Do Some Devices Get Rejected at CNEDiMTS?
Question: What are the most common reasons CNEDiMTS denies or limits device approval?
Rejection drivers: insufficient clinical evidence, weak comparator selection (fighting outdated standards instead of current practice), population too broad or misaligned with clinical reality, safety concerns unresolved, and positioning that contradicts evidence. Most preventable: poor indication strategy. Companies submit broad claims expecting broad reimbursement; instead receive narrow approval in niche populations with ASA V grading. Strategic pre-submission alignment eliminates 70% of rejections.
FAQ 5: How Does CNEDiMTS Opinion Translate to Actual Reimbursement Pricing?
Question: Does a positive CNEDiMTS opinion guarantee my device will be reimbursed and at what price?
Answer (57 words): CNEDiMTS opinion enables CEPS price negotiations—it’s foundational but not determinative. Your SA/ASA grade directly anchors pricing discussions. ASA I supports premium arguments (€50+). ASA V supports parity arguments (€30). CEPS can still negotiate below your requested price, even with a favourable CNEDiMTS opinion. Include health economics modelling in your dossier. Real-world evidence post-launch can support later price increases.
