France Redefines Diagnostic Reimbursement with the First AI-Enabled and Genomic Tests Under RIHN 2.0
Introduction
France is now setting the pace for evidence-linked reimbursement of diagnostics. Under RIHN 2.0, public funding is no longer unlocked by CE marking alone. Instead, reimbursement is earned through HAS-approved, prospective real-world evidence (RWE) that proves clinical value and health-system impact in French practice. This guide explains how to secure RIHN 2.0 reimbursement in France for AI and genomic diagnostics through HAS-approved real-world evidence.
Two decisions in 2025 made this shift unmistakable:
- 5 June 2025: HAS issued Opinion n° 2025.0027 granting conditional reimbursement to VisioCyt® Bladder (Vitadx), an AI-assisted urine cytology test for monitoring recurrence in non-muscle-invasive bladder cancer (NMIBC).
- 25 September 2025: HAS issued Opinion n° 2025.0046 granting conditional reimbursement to NOVAGRAY RILA Breast® (Novagray), a genomic radiosensitivity assay intended to personalise radiotherapy decisions in breast cancer.
Together, these rulings define the new reality of RIHN 2.0 reimbursement in France: early access is possible, but it is conditional, protocol-driven, and accountable.
Key takeaways
- RIHN 2.0 is France’s structured conditional reimbursement pathway for innovative diagnostics not yet listed in the NABM.
- Funding is tied to mandatory, HAS-approved RWE, with clear endpoints, comparators, and governance.
- AI diagnostics and genomic tests are both eligible, but must demonstrate measurable clinical and system value.
- France is aligning national reimbursement practice with European direction, including IVDR and the EU’s emerging frameworks for high-risk medical AI and joint HTA.
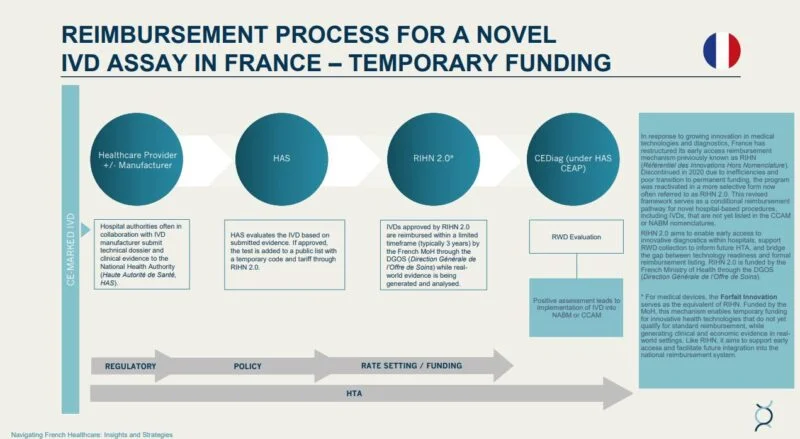
What is RIHN 2.0
RIHN 2.0 (Référentiel des Innovations Hors Nomenclature) is a national route to temporary (“prise en charge dérogatoire”) reimbursement for innovative in-vitro diagnostics (IVDs) that are not yet reimbursed via the NABM.
RIHN 2.0 is designed to bridge the gap between:
- regulatory access (e.g., CE marking under IVDR), and
- economic access (routine reimbursement and national tariff setting).
In practical terms, RIHN 2.0 pays for adoption while France learns—but only if the manufacturer agrees to generate the required evidence.
Image (RIHN pathway diagram) – recommended alt text:
RIHN 2.0 reimbursement in France: conditional funding pathway for AI and genomic diagnostics
How RIHN 2.0 works
RIHN 2.0 functions as an “evidence-for-access” contract between innovators and the health system.
1) Conditional reimbursement
Funding is granted temporarily for a defined period, typically aligned with the evidence plan.
2) A mandatory RWE protocol
A RIHN 2.0 submission must include a prospective protocol accepted by HAS. This protocol specifies:
- comparators and standard of care in France
- endpoints (clinical, organisational, economic)
- sample size and sites
- analysis approach and reporting
- data access, governance, and disclosure commitments
3) A structured transition to permanent reimbursement
If the evidence demonstrates sustained value, the diagnostic may progress to full listing in the NABM (or to other catalogue routes where relevant).
4) Joint governance (HAS + system stakeholders)
RIHN 2.0 is structured so that evidence, cost, and health-system priorities remain aligned across national bodies (HAS and relevant system governance partners).
Why CE marking is no longer enough in France
In diagnostics, CE marking establishes regulatory validity and safety requirements. It does not guarantee reimbursement.
RIHN 2.0 makes the French position explicit:
- market access is earned through French evidence, not assumed through European conformity alone
- reimbursement depends on measured pathway impact (avoidance of procedures, workload effects, downstream costs, patient outcomes)
For AI diagnostics, France’s direction is especially clear: the system expects visibility of algorithm role, data provenance, governance, and ongoing performance.
Case 1: VisioCyt® Bladder (AI urine cytology, NMIBC)
HAS Opinion n° 2025.0027 (5 June 2025) granted conditional reimbursement to VisioCyt® Bladder, positioning it as a landmark for AI-enabled IVD funding.
What the test does
VisioCyt® Bladder applies AI-assisted analysis to urine cytology to improve detection and standardisation in NMIBC surveillance.
Why it matters clinically
In NMIBC, missed recurrence can delay intervention and increase risk. An AI-supported approach is positioned to improve sensitivity and reduce variability in interpretation.
Why it matters economically
RIHN 2.0 is designed to capture not only accuracy, but system value. For NMIBC surveillance, plausible value mechanisms include:
- fewer invasive procedures
- fewer missed high-grade recurrences
- reduced laboratory burden
- improved workflow throughput in cytopathology
The RWE protocol under RIHN 2.0 is the mechanism that converts these hypotheses into auditable economic evidence.
Case 2: NOVAGRAY RILA Breast® (genomic radiosensitivity)
HAS Opinion n° 2025.0046 (25 September 2025) granted conditional reimbursement to NOVAGRAY RILA Breast®, demonstrating that RIHN 2.0 also supports precision genomic diagnostics, not only AI.
What the test does
RILA Breast® is presented as a genomic assay intended to estimate radiosensitivity and help personalise radiotherapy planning.
Why it matters clinically and economically
If a test can reduce avoidable toxicity or support better stratification of treatment intensity, the system value is not just clinical benefit but also:
- reduced management of complications
- more efficient radiotherapy resource use
- improved pathway predictability and cost control
As with AI diagnostics, the key is that these claims must be proven under a defined French protocol.
AI-specific expectations: transparency, governance, and ongoing performance
RIHN 2.0 is particularly consequential for AI because it converts “AI governance” from an ethical aspiration into a reimbursement requirement.
A strong RIHN 2.0 AI dossier typically includes:
- algorithm role and intended clinical consequence
- training/validation summary and external validation logic
- update policy and version control
- drift monitoring approach
- bias and representativeness strategy
- data governance and hosting alignment (GDPR / HDS where relevant)
RIHN 2.0 reimbursement in France: evidence requirements and strategy
Securing RIHN 2.0 reimbursement in France requires more than demonstrating technical performance or regulatory compliance. Under the renewed framework, reimbursement is explicitly conditional on the generation of prospective, HAS-approved real-world evidence (RWE) that demonstrates clinical utility, organisational relevance, and economic value within French clinical practice.
At its core, RIHN 2.0 reframes reimbursement as a contractual evidence pathway. Manufacturers are granted time-limited public funding in exchange for a clearly defined evidence-generation commitment, evaluated against endpoints that matter to patients, clinicians, laboratories, and payers.
Evidence pillars required by HAS
A successful RIHN 2.0 dossier is structured around four interdependent evidence pillars, each of which must be addressed explicitly in the submission.

1. Clinical utility (patient-level benefit)
Applicants must demonstrate that the diagnostic delivers a measurable improvement over the French standard of care, as defined in the NABM or prevailing clinical pathways. This includes clear justification of comparators, clinically relevant endpoints, and evidence that the test meaningfully influences diagnosis, monitoring, or treatment decisions.
HAS places particular emphasis on avoiding false reassurance or delayed intervention, especially in oncology and other high-risk settings, where diagnostic accuracy directly affects outcomes.
2. Organisational and workflow impact
Beyond accuracy, HAS expects evidence that the diagnostic can be implemented safely and efficiently within real laboratory and hospital environments. This includes assessment of turnaround times, reporting standardisation, staffing implications, and feasibility across multiple French sites.
For AI-enabled diagnostics, this pillar also captures the extent to which automation reduces variability, supports workforce capacity, or mitigates specialist shortages.
3. Economic and system value
RIHN 2.0 reimbursement in France is inseparable from economic accountability. Submissions must articulate a credible health-economic logic showing how clinical gains translate into system-level value. Typical mechanisms include avoided procedures, reduced downstream complications, improved throughput, or better allocation of constrained resources.
While full cost-utility analyses are not always required at entry, HAS expects a payer-credible budget impact rationale that can later support permanent tariff setting.
4. Data governance and methodological robustness
All RIHN 2.0 applications must comply with French and EU requirements on data protection, transparency, and methodological quality. RWE protocols must define governance arrangements, data access, analytical methods, and publication commitments in advance.
For AI-based diagnostics, HAS expects explicit documentation of the algorithm’s role, validation approach, update policy, and safeguards against bias or performance drift.
Strategic implications for innovators
From a strategic perspective, RIHN 2.0 reimbursement in France rewards developers who treat evidence as a core market-access asset, not a post-market obligation. The most competitive dossiers are those that align clinical endpoints, operational metrics, and economic hypotheses into a single, coherent RWE strategy.
In practice, this means designing studies that not only confirm diagnostic performance, but also explain why the test changes care pathways, how it reduces system friction, and where economic value is realised. Technologies that succeed under RIHN 2.0 are those that can demonstrate performance, relevance, and accountability simultaneously.
Strategy playbook: how to secure RIHN 2.0 reimbursement in France
If you want the single best rule:
RWE is the product. CE marking opens the door; French evidence gets you paid.
1) Design the RWE protocol early
Don’t treat the RWE protocol as post-market paperwork. Build it as a core commercial asset.
2) Make the dossier NABM-literate
Describe the current standard of care using the language of French reimbursement, and show exactly where your test changes the pathway.
3) Build governance “by design”
For AI and data-enabled diagnostics, governance is a technical requirement. Make compliance and traceability credible and auditable.
4) Quantify system value in a French payer logic
Explicitly model:
- avoided procedures
- avoided complications
- throughput gains
- workforce constraints
- downstream cost offsets
Then define which endpoints in the RWE protocol will validate them.
5) Plan the exit: RIHN 2.0 → permanent listing
RIHN 2.0 is not the finish line. Your dossier should explain the intended route to routine reimbursement and what evidence will support it.
Why this matters beyond France

France has created a practical model for conditional reimbursement of next-generation diagnostics that is compatible with Europe’s direction of travel: stronger evidence expectations, lifecycle accountability, and clearer links between value and payment.
If you can win under RIHN 2.0 reimbursement in France, you are building a template that will increasingly translate to other EU markets.
FAQs (use FAQ schema for 6 questions)
What is RIHN 2.0 in France?
RIHN 2.0 is France’s national conditional reimbursement pathway for innovative diagnostics not yet listed in the NABM, funding access while HAS-approved RWE is generated.
How do you secure RIHN 2.0 reimbursement in France?
You secure RIHN 2.0 by submitting a HAS-compliant dossier including a prospective RWE protocol demonstrating clinical utility, organisational value, and payer-relevant economic impact in France.
Does CE marking guarantee reimbursement in France?
No. CE marking supports regulatory access, but RIHN 2.0 requires nationally generated evidence to unlock conditional reimbursement.
Are AI diagnostics eligible for RIHN 2.0?
Yes. AI-enabled IVDs can be eligible, but must address transparency, governance, performance monitoring, and measurable clinical and system value.
Are genomic tests eligible for RIHN 2.0?
Yes. The NOVAGRAY RILA Breast® opinion shows RIHN 2.0 can support precision genomic assays under the same evidence-linked funding logic.
How do you move from RIHN 2.0 to permanent reimbursement?
You transition by delivering the agreed RWE outputs and demonstrating sustained value consistent with HAS expectations, enabling later progression to NABM listing (or other national catalogue routes where appropriate).
n Vitro Diagnostic Medical Devices (IVDR)
Defines analytical performance, clinical performance, and post-market evidence obligations for IVDs in the EU.
Glossary of Key Terms
Service Attendu (SA): Expected clinical/public-health benefit guiding HAS reimbursement decisions. Keywords: service attendu HAS.
AI Diagnostic (Diagnostic fondé sur l’IA): In vitro or software-as-medical-device using machine learning for data analysis; assessed by HAS for transparency and clinical utility. Keywords: AI diagnostics France, digital pathology.
Article 51 (LFSS 2018): Framework for experimental payment and care pathways enabling bundled or value-based reimbursement. Keywords: Article 51 France, value-based healthcare.
ARS (Agences Régionales de Santé): Regional health agencies coordinating innovation pilots and data collection sites. Keywords: ARS France, innovation pilot health.
CCAM: National catalogue of medical procedures; basis for coding and cost weighting. Keywords: CCAM codes France.
CNAM: National Health Insurance Fund integrating RIHN 2.0 and Forfait Innovation budgets. Keywords: CNAM reimbursement, France.
CNEDiMTS: The HAS committee for devices/IVDs evaluates RWE protocols and issues opinions. Keywords: HAS device evaluation.
CNIL / RGPD (GDPR): French data authority / EU privacy law ensuring consent, DPIA, and lawful processing. Keywords: GDPR, health data, France.
HDS: “Hébergeur de Données de Santé”; certification required for compliant data hosting. Keywords: HDS certification France.
HTA (Health Technology Assessment): Evaluation of medical, economic, and ethical dimensions guiding reimbursement. Keywords: HTA, France, EU, JCA.
IVD / IVDR: In vitro diagnostic device regulated under IVDR (EU 2017/746). Keywords: IVDR compliance, France.
LPPR / LPP: Reimbursable devices catalogue (post-Forfait Innovation integration). Keywords: LPPR France reimbursement.
Mon Espace Santé / Ségur du Numérique: National EHR infrastructure enabling interoperability and telemonitoring. Keywords: digital health, France, EHR.
NABM: Laboratory acts nomenclature; end-state for permanent reimbursement. Keywords: NABM listing France.
PECAN: Digital-health fast-track for telemonitoring / DTx / AI-assisted care. Keywords: PECAN, France, digital health.
RIHN 2.0: Conditional reimbursement for innovative IVDs linked to an HAS-approved RWE study. Keywords: RIHN 2.0 France reimbursement.
RWE (Données en vie réelle): Evidence from clinical practice on safety, performance, and economic impact; required for RIHN 2.0. Keywords: RWE France diagnostics.
References
Core RIHN 2.0 / HAS Sources
- Haute Autorité de Santé (HAS) — RIHN 2.0: Guide de dépôt de dossier (Version 2024)
Official methodological guidance and submission template for RIHN 2.0 applications, covering Parts I–V, evidence expectations, economic framing, and governance requirements. - Haute Autorité de Santé (HAS) — Déposer une demande au RIHN 2.0
Official HAS guidance detailing eligibility criteria, evaluation principles, admissible evidence, and procedural steps for conditional reimbursement under RIHN 2.0. - HAS – SESAME Portal
Secure digital platform for submission, review, and lifecycle tracking of RIHN 2.0 dossiers and HAS opinions. - HAS Opinion n° 2025.0027 — VisioCyt® Bladder (5 June 2025)
First AI-enabled in vitro diagnostic granted conditional reimbursement under RIHN 2.0, establishing precedent for AI-based diagnostics in urology. - HAS Opinion n° 2025.0046 — NOVAGRAY RILA Breast® (25 September 2025)
HAS opinion granting RIHN 2.0 access to a genomic radiosensitivity assay, reinforcing RIHN 2.0 as a structured early-access pathway for precision diagnostics.
Complementary French Funding & Policy Instruments
- Ministère de la Santé — Forfait Innovation
National early-access funding mechanism supporting innovative medical devices and procedures while clinical and economic evidence is generated. - Légifrance — Loi de Financement de la Sécurité Sociale (LFSS) 2018, Article 51
Legal framework enabling experimental payment and care-delivery models within the French health system. - Direction de la Recherche, des Études, de l’Évaluation et des Statistiques (DREES) — Évaluation des expérimentations Article 51 (2023 update)
Methodological framework defining objectives, evaluation criteria, and impact assessment for Article 51 experiments.
Reimbursement & Nomenclature Frameworks
- Assurance Maladie (Ameli) — Nomenclature des Actes de Biologie Médicale (NABM)
Official catalogue of reimbursable laboratory tests and diagnostic acts in France. - Assurance Maladie (Ameli) — Classification Commune des Actes Médicaux (CCAM)
National procedural coding system for hospital and outpatient medical acts. - Assurance Maladie (Ameli) — Liste des Produits et Prestations Remboursables (LPPR)
Catalogue governing reimbursement of medical devices and associated services.
Data Governance, Ethics & Health Data Use
- Commission Nationale de l’Informatique et des Libertés (CNIL) — Guide Santé (RGPD)
Authoritative guidance on GDPR compliance, patient consent, and data protection for healthcare and medical AI solutions. - Agence du Numérique en Santé (ANS) — Hébergeurs de Données de Santé (HDS)
Official registry of certified health-data hosting providers required for handling sensitive healthcare data in France. - Health Data Hub — SNDS Documentation (LPP / NABM)
Technical and governance documentation for access, linkage, and secondary use of national health insurance data.
European Regulatory & HTA Frameworks
- European Union — EU Artificial Intelligence Act (2024)
Regulatory framework establishing transparency, risk classification, and oversight requirements for high-risk medical AI systems. - European Parliament & Council — Regulation (EU) 2017/746 on In Vitro Diagnostic Medical Devices (IVDR)
Defines analytical performance, clinical performance, and post-market evidence obligations for IVDs in the EU. - European Union — EU HTA Regulation (EU) 2021/2282
Establishes joint clinical assessment methodologies and evidence standards for health technologies across EU Member States.