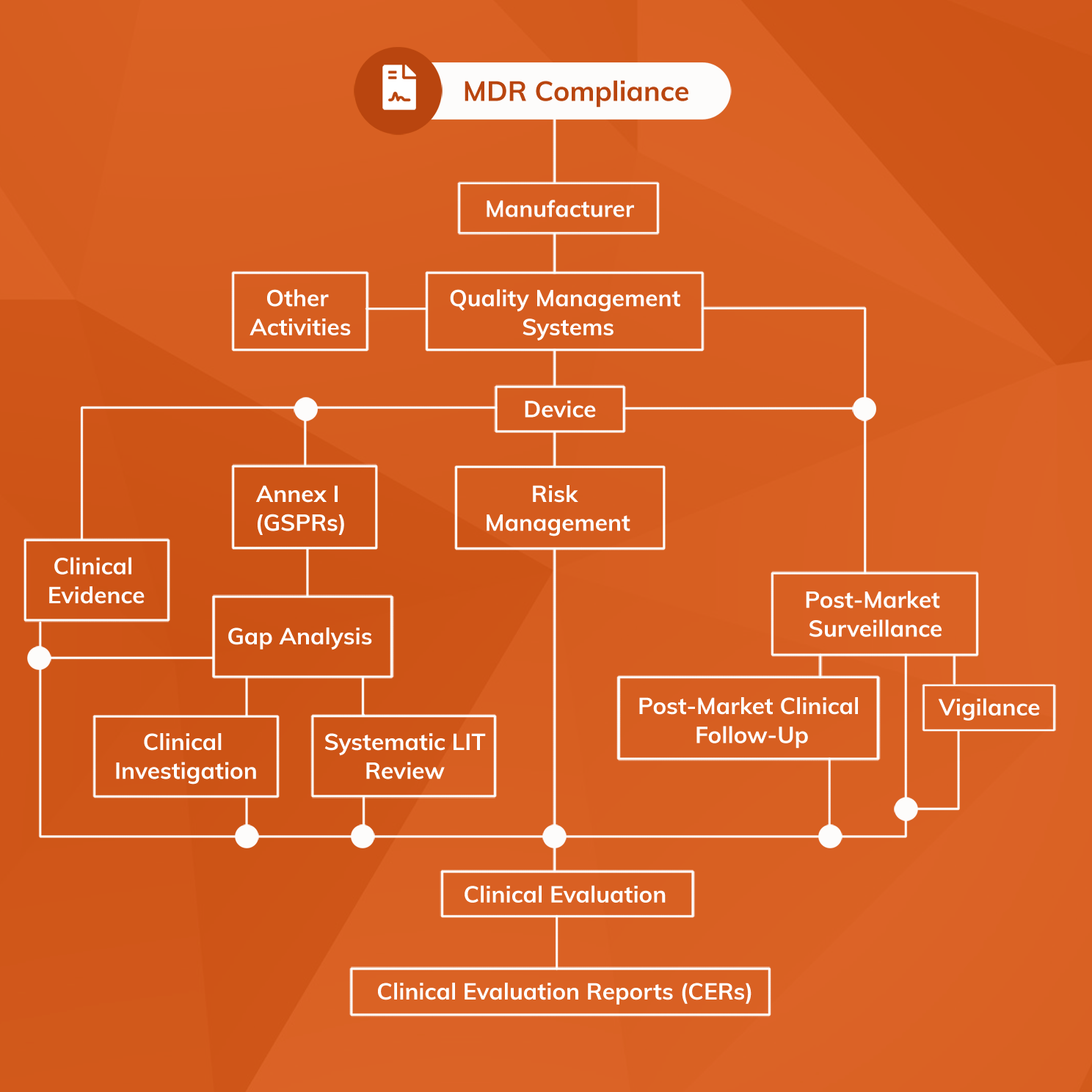
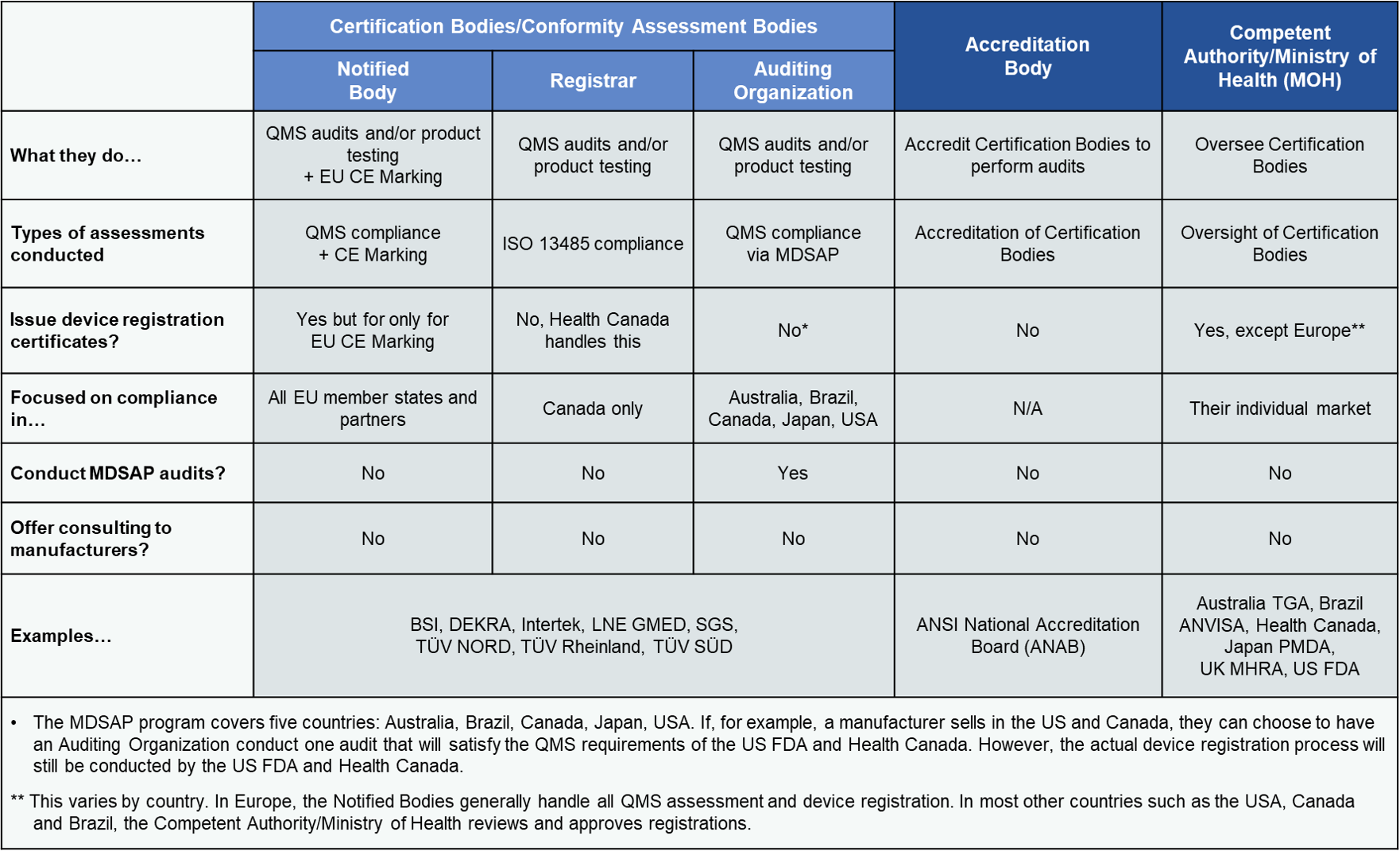
Beginning in 2026, PMCF stops being a compliance afterthought and becomes the single biggest determinant of CE-mark renewal.
Notified Bodies (NBs) are raising scientific standards, CORE-MD has formalised acceptable methodologies, and France has tightened ANSM–CNIL–CPP oversight for all post-market studies.
This guide explains how manufacturers can secure PMCF success, avoid NB refusals, and build a defensible, evidence-based lifecycle programme that satisfies MDR 2017/745 and supports reimbursement (LPPR, RIHN 2.0, HTA).
PMCF in 2026: Why the Rules Have Tightened and Why Weak PMCF Fails
By 2026, PMCF is no longer surveillance; it is continuous, testable science embedded into Annex XIV of the MDR.
NBs now reject PMCF plans that rely on phrases such as “ongoing monitoring,” “literature review,” or “routine follow-up.”
What changed?
- EU Commission 2025 NB survey: 51/51 NBs participated.
Result: near-zero tolerance for descriptive PMCF. - CORE-MD 2025 (Lancet Regional Health – Europe): formally defines the minimum scientific expectations.
- Team-NB 2025 BPG v3: establishes strict evidence traceability across CER → PMCF Plan → PMCF Evaluation Report → PSUR → Risk.
Weak PMCF = high risk of major NB nonconformities, certification conditions, or refusal.
How to Secure PMCF: What Every Manufacturer Must Demonstrate in 2026
To pass NB review, PMCF must now prove five non-negotiable scientific elements:
1) Clear, testable hypotheses tied to CER uncertainties
Every PMCF must explicitly answer:
Which residual risks or uncertain performance parameters remain unresolved pre-CE?
2) Quantifiable and clinically meaningful endpoints
NBs expect validated clinical endpoints such as:
- Diagnostic AUC / calibration
- Revision-free survival
- EQ-5D-5L or PROMIS
- Device function over time
- Adverse event incidence
3) A fully documented Statistical Analysis Plan (ISO 14155 aligned)
Must include:
- Power ≥ 80–90%
- Effect size justification
- Missing-data strategy
- Confounder control (propensity scores, multivariable models)
- Pre-specified subgroup analyses
4) Data provenance, governance, and GDPR compliance
In France this means explicit CNIL MR-001 compliance and clear data lineage.
5) Lifecycle feedback loops
PMCF must update:
- CER
- PSUR
- IFU
- Risk Management (ISO 14971)
Without these, NBs increasingly issue NCs.
How to Design PMCF That Meets 2026 NB Expectations (Practical Blueprint)
Below is the structure that consistently secures NB approval:
Step 1 Map CER residual uncertainties → PMCF objectives
Examples:
- Risk of late device failure
- Variability across operators or sites
- Real-world diagnostic performance vs controlled settings
- Off-label use or misuse patterns
Step 2 Select a defensible study design
| Design | When it secures NB approval | Why NBs prefer it (CORE-MD 2025) |
|---|---|---|
| Prospective observational | Class IIb/III, or any device with performance uncertainties | Transparent, real-world, reproducible |
| Registry-embedded PMCF | Multi-country or high-risk devices | Gold-standard for RWE; scalable; generalisable |
| Retrospective EMR PMCF | Signal verification or early confirmation | Fast + cheap; acceptable if bias-controlled |
| Interventional PMCF (France Art. 74(1)) | Added imaging/biopsy, additional visits | Required when procedures exceed routine care |
NB reviewers increasingly ask:
Why is this design the minimum scientifically sufficient option?
Your plan must answer this.
Step 3 Build the Statistical Analysis Plan
Include:
- Power calculation
- Covariate selection
- Missing data handling
- Sensitivity analyses
- Pre-defined subgroup analyses
- Multiplicity control when necessary
This is what makes PMCF scientific, not administrative.
Step 4 Data governance (2026 priority)
For France (mandatory text to include in PMCF Plan):
- CNIL MR-001 compliance
- Hosting on HDS-certified EU servers
- Data minimisation + pseudonymisation
- Role-based access controls
- Use of the Health Data Hub (HDH) 2025 Toolkit for SNDS use
NBs now explicitly check data lineage diagrams.
Step 5 Lifecycle integration
PMCF must clearly show how findings:
- Update CER
- Update PSUR frequency and content
- Trigger CAPA
- Modify the IFU
- Reduce uncertainties for the next CE renewal
This is the heart of MDR Annex XIV.
How France’s Rules Affect PMCF Success (2026 Version)
France remains the strictest EU country for PMCF due to combined regulatory–ethical–data oversight.
Case 3 Article 74(1) MDR (Interventional PMCF)
Requires:
- ANSM approval
- CPP ethics approval
- CNIL compliance (MR-001)
- Insurance
Triggers:
- Added imaging
- Additional blood samples
- Extra hospital visits
- Procedures beyond routine care
Case 4.1 Article 82 (Non-interventional PMCF)
Requires:
- CPP
- CNIL MR-001
No ANSM; no insurance.
Retrospective PMCF (Data-only)
Requires:
- CESREES
- CNIL
Route via HDH for SNDS access.
France adds 2–3 months minimum to PMCF timelines → build this into CE-renewal schedules
Securing PMCF with RWE: What NBs Now Accept
The EMA’s 2025 report (59 regulator-led RWE studies) legitimised RWE for lifecycle decisions.
NBs are aligning rapidly.
For PMCF, RWE is acceptable only when it demonstrates:
✔ Clear data provenance
✔ Bias mitigation (new-user design, negative controls, PS weighting)
✔ Device–outcome linkage (UDI → registry → outcomes)
✔ Clinically validated endpoints
✔ Auditable data access (HDH/EHDEN/DARWIN EU standards)
If your PMCF Plan includes these, NB reviewers treat RWE as scientific evidence, not “supporting material.”
The 2026 PMCF Quality Standard (What NBs Actually Want to See)
Below is the exact checklist NBs use in 2026 (derived from NB survey data, Team-NB BPG v3, CORE-MD):
Scientific Quality
✓ Hypothesis-driven
✓ Validated endpoints
✓ SAP included
✓ Adequate power
✓ Comparator justified
✓ Predefined subgroups
Data Quality
✓ GDPR compliant
✓ CNIL MR-001 (France)
✓ De-identification strategy
✓ Data lineage diagram
✓ EU-based hosting
Lifecycle Traceability
✓ CER → PMCF Plan → PMCF Evaluation Report → PSUR → Risk Management
✓ CAPA triggers
✓ IFU update policy
Regulatory Alignment
✓ Article 74/82 classification justified
✓ Ethics approvals clear and realistic
✓ Multi-country governance plan
If any one of these is missing → NB issues nonconformity.
The 2026 PMCF Playbook: How to Build a Passing Dossier
To secure PMCF success:
1. Start with CER gaps not with a study design
NBs reject designs that don’t close residual uncertainties.
2. Use registry-embedded PMCF wherever possible
CORE-MD identifies this as the gold standard.
3. Build a real SAP not a paragraph
ISO 14155 alignment is now mandatory in practice.
4. Embed data governance in the PMCF Plan
CNIL MR-001 must be visible.
5. Pre-write your PMCF Evaluation Report structure
NBs expect to see how results will feed CER & PSUR.
6. Build PMCF with HTA reuse in mind
Endpoints like EQ-5D-5L, revision-free survival, complications → support reimbursement.
7. Use OMOP mapping if future RWE is expected
Accelerates reuse in EHDEN, DARWIN EU, and payer submissions.
8. Document everything traceability is now the deciding factor
NBs expect audit-ready evidence.
FAQ
Here are your FAQs rewritten to align with the new blog title + SEO intent.
(I can provide the full expanded FAQ set if you want.)
Q1. What is the purpose of PMCF under MDR — and what secures NB approval?
PMCF verifies ongoing safety, performance, and clinical benefit using quantitative, hypothesis-driven evidence.
NBs approve PMCF when it clearly reduces CER uncertainties and provides traceable, reproducible clinical data.
Q2. What changed in PMCF expectations for 2026?
NBs now require:
- A full SAP
- Validated endpoints
- Data governance documentation
- Evidence traceability across CER–PMCF–PSUR–Risk
The 2025 NB survey confirms that descriptive PMCF is no longer accepted.
Q3. What study designs secure the fastest NB acceptance?
- Registry-embedded PMCF
- Prospective observational cohorts with clear endpoints
- Retrospective EMR studies (if bias-controlled)
Interventional PMCF (France) is required only when additional procedures exceed routine care.
Q4. How do I secure PMCF approval in France?
Correct classification is critical:
- Article 74(1) → ANSM + CPP + CNIL + insurance
- Article 82 → CPP + CNIL
- Retrospective → CESREES + CNIL via HDH
Incorrect classification is a common cause of delays.
Q5. How does PMCF support reimbursement?
PMCF produces real-world, quantitative outcomes that populate:
- LPPR/RIHN 2.0 submissions (France)
- HTA dossiers (EU)
- Cost-consequence and budget impact models
NBs, HTA bodies, and payers now converge around the same evidence principles.
PMCF in 2026 Is Winnable, But Only With Scientific Discipline
PMCF is no longer a formality; it is the centrepiece of EU MDR evidence.
Manufacturers that design PMCF as living clinical science — instead of surveillance — will:
- Renew CE marks smoothly
- Reduce NB queries
- Strengthen HTA and reimbursement cases
- Build long-term regulatory defensibility
The path to securing PMCF is clear:
Hypotheses → endpoints → SAP → governance → lifecycle integration → registry or RWE extension.
References
1. Dobrzynska et al. NB use of conditional certification
Citation (
Dobrzynska, A., Rejón-Parrilla, J.C., Epstein, D., Aranda-López, J., Fraser, A.G. and Blasco-Amaro, J.A. (2025) ‘Survey of notified bodies reveals very limited use of conditional certification for high-risk medical devices’, Frontiers in Medical Technology, 7, 1504294. doi:10.3389/fmedt.2025.1504294. Survey of Notified Bodies reveals very limited use of conditional certification for high-risk medical devices – PubMed
What it shows
CORE-MD surveyed 13 NBs and found that only a small minority used conditional certification for high-risk devices, confirming that “approve now, prove later” is the exception, not the rule. This is your core evidence that NBs now expect robust PMCF and CER at the time of certification, not deferred evidence.
2. Fraser et al. CORE-MD methodological framework
Citation (Harvard)
Fraser, A.G., Butchart, E.G., Schlander, M. et al. (2025) ‘Recommended methodologies for clinical investigations of high-risk medical devices: Conclusions from the EU CORE-MD project’, The Lancet Regional Health – Europe, 42, 101460. doi:10.1016/j.lanepe.2025.101460. Recommended methodologies for clinical investigations of high-risk medical devices—Conclusions from the European Union CORE–MD Project – The Lancet Regional Health – Europe
What it shows
This CORE-MD consensus defines a hierarchy of evidence for high-risk devices: registry-embedded studies, pragmatic RCTs (including ethically acceptable sham controls), and transparent SAPs as the new benchmark for EU device trials. It’s the main scientific backbone for your claims about PMCF design, registry-based trials, and staged evidence frameworks.
3. European Commission Notified Bodies Survey 2025
Citation
European Commission, Directorate-General for Health and Food Safety (2025) Update – Notified bodies survey on certifications and applications, 15 May 2025. Update – Notified bodies survey on certifications and applications – Public Health
What it shows
Annual NB monitoring with 51/51 NBs responding; the report quantifies MDR certification volumes, application backlogs, and NB capacity. It supports your macro-statement that NB oversight is now systematic and that expectations around clinical evidence (including PMCF) are harmonised and closely monitored.
4. EMA Regulator-led RWE report (59 studies)
Citation
European Medicines Agency (2025) Real-world evidence: report on regulator-led studies 2024–2025, European Medicines Agency, Amsterdam. La Team NB publie la V3 de son guide pour les dossiers techniques MDR : des nouveautés à ne pas ignorer !
What it shows
The EMA summarises 59 regulator-led or regulator-initiated RWE studies completed or ongoing between Feb-2024 and Feb-2025, showing a ~47% increase vs the prior year. It’s your key evidence that curated RWD/RWE is now mainstream in EU lifecycle decision-making and that PMCF aligned with RWE methods is regulator-credible.
5. Team-NB Best Practice Guidance v3 (Technical Docs)
Citation
Team-NB (2025) Best Practice Guidance for Technical Documentation under EU MDR – Version 3, Team-NB, Brussels, April 2025. Team-NB-PositionPaper-BPG-TechnicalDocEU-MDR-2017-745-V3-20250409.pdf
What it shows
This guidance from the NB association explains what NBs expect in technical documentation: traceability between CEP/CER, PMCF Plan, PSUR and Risk Management, plus clearer SAPs and justification of clinical endpoints. It underpins your statements on document cohesion and “living” evidence systems.
6. Health Data Hub (France) CNIL/CESREES/SNDS toolkit
Citation (
Health Data Hub (2025) Starter kit et check-lists pour les demandes d’autorisation CNIL et l’accès au SNDS, Health Data Hub, Paris. Réaliser sa demande d’autorisation auprès de la CNIL | Health Data Hub
What it shows
HDH’s “starter kit” and associated toolkits explain how to prepare CESREES/CNIL applications, how simplified procedures work, and how to access SNDS datasets and aggregates. This is your main reference for the “France playbook” on retrospective PMCF, registry/RWD studies, and realistic timelines (~2–3 months for approvals).
7. MDCG 2020-7 PMCF Plan template
Citation
Medical Device Coordination Group (2020) MDCG 2020-7: Post-market clinical follow-up (PMCF) plan template – A guide for manufacturers and notified bodies, European Commission, Brussels.
What it shows
Defines the mandatory structure and content of a PMCF Plan (objectives, methods, endpoints, timelines, rationale for activities or justified non-use). This is the formal regulatory anchor for your blueprint section (objectives, endpoints, design, statistics, feedback loops).
Link
Official MDCG 2020-7 PMCF Plan template (DocsRoom)
8. MDCG 2020-8 PMCF Evaluation Report template
Citation
Medical Device Coordination Group (2020) MDCG 2020-8: Post-market clinical follow-up (PMCF) evaluation report template – A guide for manufacturers and notified bodies, European Commission, Brussels.Microsoft Word – 08 Guidance on PMCF Evaluation Report Template
What it shows
Sets out how PMCF results must be reported back into the technical file, including summary of methods, data, results, benefit–risk reassessment and implications for IFU and risk management. It’s your reference when you describe PMCF Evaluation Reports feeding the CER and PSUR.
9. CNIL MR-001 reference methodology
Citation
Commission Nationale de l’Informatique et des Libertés (CNIL) (2025) Méthodologie de référence MR-001: Recherches dans le domaine de la santé avec recueil du consentement, CNIL, Paris (updated). Google v. CNIL: EU Rules that Right to be Forgotten Does Not Apply Globally – Harvard Journal of Law & Technology
What it shows
MR-001 is the core French GDPR framework for health research that involves consented participants. It defines legal bases, data-minimisation, retention, security, and roles for investigators, statisticians and monitors—essential for any French PMCF (prospective, interventional or non-interventional) involving identifiable data.
10. ANSM — Article 74(1) clinical investigations
Citation
Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM) (2025) Investigations cliniques de dispositifs médicaux : classification et process d’évaluation selon le règlement (UE) 2017/745 (RDM), updated 28 March 2025. Accueil – ANSM
What it shows
ANSM’s guidance and classification tables clarify when a PMCF becomes an “investigation clinique” under Article 74(1) MDR and how France applies additional national rules. This is your legal basis for describing “Case 3” (CE-marked + invasive/burdensome → ANSM + CPP + CNIL + insurance).
11. CPP — Ethics committees for research involving humans
Citation
Ministère de la Santé / Commission nationale des recherches impliquant la personne humaine (CNRIPH) (2025) La commission nationale des recherches impliquant la personne humaine: missions et fonctionnement, Ministère de la Santé, Paris. La Commission nationale des recherches impliquant la personne humaine – CNRIPH – Ministère de la Santé, de la Famille, de l’Autonomie et des Personnes handicapées
What it shows
Explains the national commission’s role in coordinating CPPs (Comités de Protection des Personnes), which issue ethics opinions for French biomedical research, including PMCF under Articles 74 and 82 MDR. It underpins your statements about CPP approvals in both interventional and non-interventional PMCF.
12. CESREES Ethics for data-only / retrospective studies
Citation
CESREES / Health Data Hub (2025) Comité éthique et scientifique pour les recherches, les études et les évaluations dans le domaine de la santé (CESREES): missions, composition et calendrier, Health Data Hub, Paris. Health Data Hub
What it shows
Describes CESREES’ remit and processes for reviewing secondary-use health-data projects (including retrospective PMCF using SNDS/registries). It supports your description of “retrospective/data-only” PMCF falling under CESREES + CNIL rather than MDR Article 74/82.
Link
CESREES – Comité éthique et scientifique pour les recherches, les études et les évaluations dans le domaine de la santé | Documentation du SNDS & SNDS OMOP
13. SNDS National Health Data System portal
Citation
Système National des Données de Santé (2025) Portail du Système national des données de santé (SNDS), Caisse Nationale d’Assurance Maladie, Paris. Accueil | SNDS
What it shows
The SNDS portal explains what the SNDS is, how access procedures work, and how data from multiple national sources (claims, hospitalisations, mortality, disability) are linked. This is your anchor when you talk about SNDS as a backbone for retrospective PMCF and large-scale RWE in France.
14. EHDEN OMOP and federated EU RWE network
Citation
European Health Data & Evidence Network (EHDEN) Consortium (2018–2025) European Health Data & Evidence Network: federated OMOP-mapped data partners in Europe, EHDEN, Brussels. IHI Innovative Health Initiative+3ehden.eu+3documentation-snds.health-data-hub.fr+3
What it shows
EHDEN is the main EU-funded project mapping hundreds of European data sources to the OMOP common data model within a federated network. It gives you a concrete basis for recommending OMOP-aligned PMCF databases and for your “design for multi-country reuse” / RWE interoperability messaging.
Key links
EHDEN project portal
EHDEN overview (IHI factsheet)
15. DARWIN EU EMA’s federated RWE platform
Citation
European Medicines Agency (2025) DARWIN EU® – Data Analysis and Real-World Interrogation Network, European Medicines Agency, Amsterdam. European Medicines Agency (EMA)+1
What it shows
DARWIN EU is EMA’s federated network of European data partners delivering RWE to support regulatory decisions on medicines (and, by extension, methods for device-relevant outcomes). You use it to show that federated, OMOP-aligned RWE infrastructures are now embedded in EU regulation—strengthening the logic for registry-embedded PMCF and cross-country data reuse.
Key links
EMA page on DARWIN EU
Dedicated DARWIN EU site
